Concentration Calculation From UV Vis Absorbance
Concentration Calculation From UV Vis Absorbance - InstaNANO. https://instanano.com/all/characterization/uv-vis/concentration/ (accessed April 15th, 2024).
This calculator can be used to determine the concentration of any sample by using absorbance and molar absorptivity values.
Absorbance (a.u.)
Molar Absorptivity (L/mmol.cm)
Cell Length (cm)
Calculation Tutorial:
STEP1:Open the absorbance graph of the solution, which is obtained from the UV Vis spectroscopy.
STEP2: Now zoom on the peak for which you want to calculate the concentration and note down the Absorbance value.
STEP3: Now enter the measured absorbance value (eg. 0.84) into the “Absorbance of Solution” column of the calculator; also enter the value of “Molar Absorptivity” (eg. 19400) of that material from the literature survey and “Solution Cell Length” (eg. 1 cm) in the next columns. You should get the calculated results of the band gap in the “Calculated Result” field.
NOTE: For accuracy you can make a solution for known concentration and enter the values of “Absorbance of Solution”, “Solution Cell Length” and “Known Concentration of Solution” in the given columns. You should get the value of “Molar Absorptivity” from the calculator.
Theory Behind Calculations:
Calculations are based on the Beer’s Lambert law given by:
A = ε l c
Where A = Absorbance of solution at a particular wavelength; ε = Molar Absorptivity; l = Length of Solution Cell; and c = Concentration of Solution (mol/dm3)






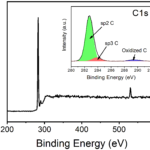
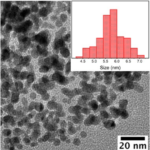
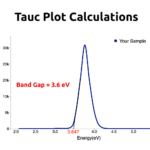


I have absorbance ( at 540nm) and reaction time
how to find the enzyme activity.
Why do I get a negative result for calculating creatinine concentration on a spectrophotometer of all serum samples
Hello. I have data for uv vis spectra of iodine in different temperatures . And i have to find the partial pressure diagram iodine. I have just temperatures and absorbances . How can i find the molar extinction coefficient at different temperatures and absorptions?