UV Vis Spectroscopy Band gap Calculator
UV Vis Spectroscopy Band gap Calculator - InstaNANO. https://instanano.com/all/characterization/uv-vis/band-gap/ (accessed April 24th, 2024).
Band gap of any material can be easily calculated by using UV-Vis absorption wavelength or lamda max value of the sample.
Calculation Tutorial:
STEP1: Open the absorption graph of the material, which is obtained from the UV Vis spectroscopy.
STEP2: Now zoom on the area for which you want to calculate the band gap and note down the wavelength (λ) at which absorption peak is shown.
STEP3: Now enter the measured wavelength value (eg. 31.8) into the ” Wavelength of Absorption peak (in nanometer)” column of the calculator. You should get the calculated results of the band gap in the “Calculated Result” field.
Theory Behind Calculations:
UV Vis Spectroscopy absorption peak means the Electrons are absorbing the Energy at some specific wavelength.
Electrons are absorbing Energy means the Electrons are going to excited state from its ground state.
Electrons are going to excited state from its ground state means the material is having band gap, thus which can be determine by absorption wavelength.
Energy Equation of Quantum Mechanics:
Energy (E) = Planks Constant (h) * Speed of Light (C) / Wavelength (λ)
Where, Energy (E) = Band gap, Planks constant (h) = 6.626×10^-34 Joules sec, Velocity of Light (C) = 2.99×10^8 meter/sec and Wavelength (λ) = Absorption peak value. Also 1eV = 1.6×10^-19 Joules (Conversion factor)
By this formula band gap can be calculated easily, from UV Vis spectroscopy absorption peak.






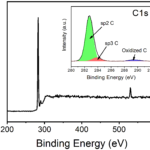
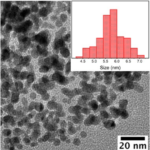
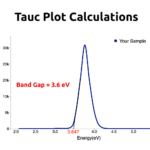


Can I get the bandgap energy of Co0.5Zn0.5Fe2O4@Polyaniline and ZnFe2O4@polyaniline compounds?
FOR CALCULATING THE BAND GAP ENERGY IT IS VERY SIMPLE AND USEFUL FOR RESEARCHERS