Copper Nanoparticles Green Synthesis by using Copper Nitrate and Bottle Brush Leaves via Biological Method
Cite This in Your Publication
Copper Nanoparticles Green Synthesis by using Copper Nitrate and Bottle Brush Leaves via Biological Method - InstaNANO. https://instanano.com/all/nanomaterial-synthesis/metal/copper-nanoparticles-2/ (accessed April 16th, 2024).
Copper Nanoparticles Green Synthesis by using Copper Nitrate and Bottle Brush Leaves via Biological Method - InstaNANO. https://instanano.com/all/nanomaterial-synthesis/metal/copper-nanoparticles-2/ (accessed April 16th, 2024).
Copper Nanoparticles Green Synthesis by using Copper Nitrate as Precursor and Bottle Brush Leaves extract as Reducing and Stabilising Agent via Biological Method.
-
CHECK LISTFresh leaves of Bottle Brush, Copper Nitrate, Deionized Water, RB Flask, Dropper, Magnetic Stirrer with Temperature option, FIlter Paper
-
STEP 1.Take fresh leaves of Bottle Brush. Wash properly and cut them into small pieces
-
STEP 2.Take 25g leaves pieces in 100ml of deionized water and boil the leaves around 80-90ºC for 5-10 minutes.
-
STEP 3.When the water turns GREEN, filter it out and use it as reducing and capping agent for Nanoparticles synthesis.
-
STEP 4.Take 0.5g Copper Nitrate in 100ml deionized water; and set the temperature 60-70ºC.
-
STEP 5.Add leaf extract drop wise slowly (prepared in step 3) into Copper Nitrate solution, until the color changes to light GREEN.
-
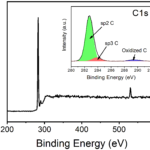
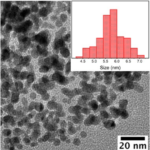
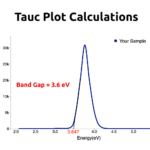
RESULTSThe light Green color indicated the formation of Copper Nanoparticles in water. UV-Vis spectroscopy absorption peak would be obtained around 600nm.
-
Factors Affecting SynthesisSpecies of Plant/Tree: Different types of Plants/Trees are having different types of chemical composition and concentration of specific chemical in its leaves. So different species of Plants/Trees can give the different particle size, size distribution. Even morphology can also be changed by using leaf extract of different plants/trees.
Concentration of Copper Nitrate: Nickel Chloride is used as the precursor in this synthesis. Higher the concentration of Nickel Chloride, more the chances of agglomeration; further leads to bulky size particles.
Concentration of Leaves in Water: Concentration of leaves in water is very important for Nanoparticles synthesis. Higher concentration of leaves in water, leads to higher concentration of reducing and capping agent in water.
Temperature: The temperature is another important factor in the synthesis of Nanoparticles, change in temperature leads to change in the particles size; also higher temperature leads to faster chemical Kinetics. -
NOTE: All the experiments should be done under the guidance of lab Incharge; and proper lab safety instructions.























Where I can get the bottle brush leaves
i have used hemi graphis coloratta leaf for extract and copper nitrate i got the colour change in brown what dose it shows presence or absence of nanopraticle formulations